A) the strong nuclear force saturates.
B) nucleons don't move throughout the nucleus.
C) all nuclei have the same volume.
D) the radius of a nucleus is directly proportional to the number of nucleons.
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The isotope 14C cannot be used in dating old samples of which of the following?
A) charcoal from a fire
B) a bronze implement from a cave
C) a bone buried in mud
D) All of the above can be dated using 14C.
F) B) and C)
Correct Answer

verified
B
Correct Answer
verified
Multiple Choice
A radioactive isotope that emits a gamma quantum will change in what respect?
A) Atomic number increases by one.
B) Atomic number decreases by one.
C) Atomic mass number decreases by one.
D) None of the above choices are valid.
F) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What particle is emitted when 20Na decays to 20Ne? (atomic numbers of Na and Ne are, respectively, 11 and 10)
A) alpha
B) beta (electron)
C) beta (positron)
D) gamma quantum
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Tritium has a half-life of 12.3 years.How many years will elapse when the radioactivity of a tritium sample diminishes to 20% of its original value?
A) 21 years
B) 29 years
C) 57 years
D) 86 years
F) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The Q value of the reaction 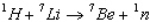 is -1.65 MeV.What is the threshold energy of this reaction? (Hint: Once the Q value is known, the relative mass numbers are precise enough to yield the result to 3 significant figures, actual masses not being needed) .
is -1.65 MeV.What is the threshold energy of this reaction? (Hint: Once the Q value is known, the relative mass numbers are precise enough to yield the result to 3 significant figures, actual masses not being needed) .
A) 1.65 MeV
B) 1.89 MeV
C) 1.77 MeV
D) 1.41 MeV
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A radioactive material initially is observed to have an activity of 1 000 decays/sec.If three hours later it is observed to have an activity of 125 decays/sec, what is its half-life?
A) 1/2 hour
B) 1 hour
C) 3 hours
D) 8 hours
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What energy must be added or given off in a reaction where two hydrogen atoms and two neutrons are combined to form a helium atom? (Atomic masses for each: hydrogen, 1.007 825 u; neutron, 1.008 665 u; helium, 4.002 602 u; also, 1 u = 931.5 MeV/c2)
A) 20.7 MeV added
B) 20.7 MeV given off
C) 28.3 MeV given off
D) 28.3 MeV added
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The binding energy of a nucleus is equal to:
A) the energy needed to remove one of the nucleons.
B) the average energy with which any nucleon is bound in the nucleus.
C) the energy needed to separate all the nucleons from each other.
D) the mass of the nucleus times c2.
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
To what is the radiation damage in biological organisms primarily due?
A) helium introduction
B) heating
C) induced radioactivity
D) ionization
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In the beta decay of 14C, the existence of the antineutrino was required to maintain:
A) energy conservation.
B) charge conservation.
C) conservation of the number of nucleons.
D) all of the above.
F) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The alpha emission process results in the daughter nucleus differing in what manner from the parent?
A) Atomic mass increases by one.
B) Atomic number decreases by two.
C) Atomic number increases by one.
D) Atomic mass decreases by two.
F) A) and C)
Correct Answer

verified
B
Correct Answer
verified
Multiple Choice
An element is emitting alpha, beta, and gamma radiation.Rank order them in terms of the thickness of protective shielding they will need for safety, from least to most.
A) alpha, beta and gamma
B) gamma, beta and alpha
C) beta, gamma and alpha
D) alpha, gamma and beta
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The mass of 238U is not quite an integer multiple of 1H mass.This is primarily because:
A) the proton and neutron have different masses.
B) there are several isotopes of uranium.
C) of the binding energy of uranium.
D) uranium is radioactive.
F) C) and D)
Correct Answer

verified
C
Correct Answer
verified
Multiple Choice
For every stable nucleus except hydrogen, if the number of nucleons is doubled, the quantity that will change least is the:
A) mass.
B) charge.
C) volume.
D) density.
F) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The atomic number of a given element is equivalent to which of the following?
A) proton number in the nucleus
B) neutron number in the nucleus
C) sum of the protons and neutrons in the nucleus
D) number of electrons in the outer shells
F) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Sample #1 is made from an isotope with decay constant 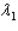 and sample #2 is made from an isotope with decay constant
and sample #2 is made from an isotope with decay constant 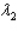 , where
, where 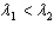 .Which of the following statements must be true?
.Which of the following statements must be true?
A) The activity of sample #1 is greater than that of sample #2.
B) The activity of sample #2 is greater than that of sample #1.
C) The half-life exhibited for sample #1 is greater than that of sample #2.
D) The half-life exhibited for sample #2 is greater than that for sample #1
F) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The isotope 64Zn has a nuclear radius of 4.8 * 10-15 m.Approximately what is the nuclear radius of the isotope 27Al?
A) 2.0 * 10-15 m
B) 2.7 * 10-15 m
C) 3.6 * 10-15 m
D) 4.0 * 10-15 m
F) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The existence of the neutrino was postulated to account for which basic conservation laws during the beta decay process?
A) conservation of energy
B) conservation of momentum
C) Both choices a and b are valid.
D) None of the above choices are valid.
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The isotope 238U, which starts one of the natural radioactive series, decays first by alpha decay followed by two negative beta decays.At this point, what is the resulting isotope?
A) (238U)
B) (236U)
C) (234Th (Z = 90 for Th.) )
D) Some other uranium isotope not given above.
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Showing 1 - 20 of 79
Related Exams