A) It has a high surface tension.
B) It is a solute.
C) Its bonds are nonpolar.
D) It can dissolve many chemical compounds.
E) It can absorb and release heat without changing temperature very much.
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
All of the synthesis reactions in the body are called
A) oxidation-reduction.
B) hydrolysis.
C) dissociation.
D) anabolism.
E) catabolism.
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Glycogen is the
A) storage carbohydrate in animals.
B) storage carbohydrate in plants.
C) sugar found in RNA.
D) major nutrient for most body cells.
E) nondigestible plant polysaccharide.
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
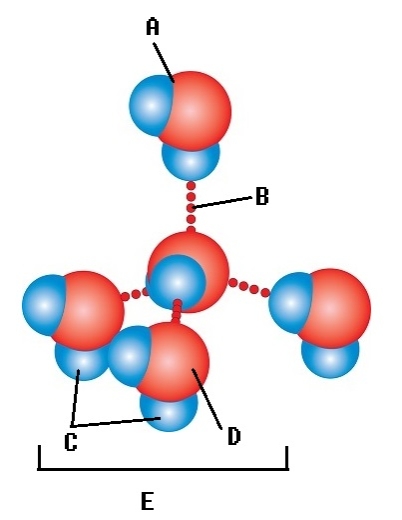 -Water accounts for 50% of the weight of a young adult female and 60% of a young adult male. What kind of atom is found at "D"?
-Water accounts for 50% of the weight of a young adult female and 60% of a young adult male. What kind of atom is found at "D"?
A) oxygen atom
B) water molecule
C) polar covalent bond
D) hydrogen atom
E) hydrogen bond
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following is a component of a nucleotide?
A) adenine--a nitrogen base
B) calcium ions
C) cholesterol--a steroid
D) ATP
E) glucose--a monosaccharide
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following factors will influence the rate of chemical reactions?
A) temperature
B) concentration of reactants
C) presence of catalysts
D) presence of enzymes
E) All of these factors will influence the rate of chemical reactions.
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The mass of a chemical equal to its molecular weight in grams, containing 6.023 x 1023 molecules is a(n)
A) atomic mass unit
B) ion
C) mole
D) molarity
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following molecules is NOT made from cholesterol?
A) testosterone
B) progesterone
C) bile salts
D) estrogen
E) prostaglandins
G) A) and C)
Correct Answer

verified
Correct Answer
verified
True/False
The importance of O2 in the human body is to extract energy (ATP) from food molecules.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A group of water molecules are held together by
A) salt.
B) double covalent bonds.
C) polar covalent bonds.
D) ionic bonds.
E) hydrogen bonds.
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What particle is formed when an acid loses a proton (H+) ?
A) conjugate base
B) buffer
C) conjugate acid
D) salt
F) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following is a carbohydrate?
A) animal fat
B) triglyceride
C) sucrose
D) cholesterol
E) hemoglobin
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Potential energy stored in bonds of molecules is _____ energy.
A) mechanical
B) thermal
C) chemical
D) molecular
E) None of the above
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
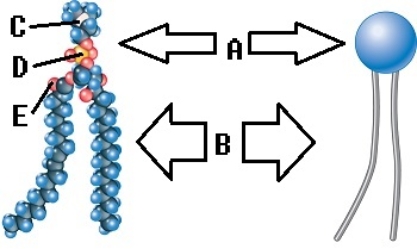 -Phospholipids are important components of the plasma membrane. What does "B" represent on the diagram?
-Phospholipids are important components of the plasma membrane. What does "B" represent on the diagram?
A) phosphorus
B) oxygen
C) nitrogen
D) polar (hydrophilic) region
E) nonpolar (hydrophobic) region
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
An organic molecule such as a vitamin that makes an enzyme functional is called a/an
A) coenzyme.
B) buffer.
C) catalyst.
D) coactivator.
F) A) and B)
Correct Answer

verified
Correct Answer
verified
True/False
Synthesis reactions are also called catabolic reactions.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following lists the components of a nucleotide?
A) phosphate-sucrose-amino acid
B) phosphate-lipid-organic base
C) monosaccharide-organic base-sucrose
D) monosaccharide-amino acid-phosphate
E) phosphate-monosaccharide-organic base
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following chemical reactions best represents the synthesis of ATP?
A) ATP + H2O ADP + Pi + energy
B) ATP + energy ADP + H2O
C) ADP + Pi + energy ATP + H2O
D) ATP + ADP ATP
E) ADP + ADP + ADP ATP + energy
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The primary structure of a protein is
A) the hydrogen bonds between amino acids.
B) the folded, helical nature of the molecule.
C) represented by multiple polypeptide chains.
D) the sequence of amino acids in the polypeptide chain.
E) the number of polypeptide chains in the molecule.
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A molecule is
A) a substance that conducts electricity when placed in solution.
B) a positively charged ion.
C) an alteration in the three-dimensional structure of a protein.
D) a combination of atoms held together by chemical bonds.
E) a negatively charged ion.
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Showing 81 - 100 of 168
Related Exams